High-entropy alloys (HEAs), which are formed by combining nearly equal parts of several – usually five or more – primary metals, are an emerging class of advanced materials that hold great potential for creating materials with superior mechanical, thermal, and catalytic properties. Another attraction of HEAs is that they can create effective alternatives to materials that are scarce, hazardous, expensive, or subject to international restrictions or conflict.
Similar to conventional alloys, industrial operation environments for HEAs usually involve critical conditions such as exposure to high temperature, oxidizing/reducing gases, and acidic and chloride-containing solutions.
Several studies have been devoted to understanding the corrosion behaviors of HEAs such as oxidation in air and oxygen-containing atmospheres. Nevertheless, there currently is little knowledge of how HEA materials, especially in nanoparticulate form, behave under critical conditions involving oxidizing gases due to the limited characterization methods available for dynamic events.
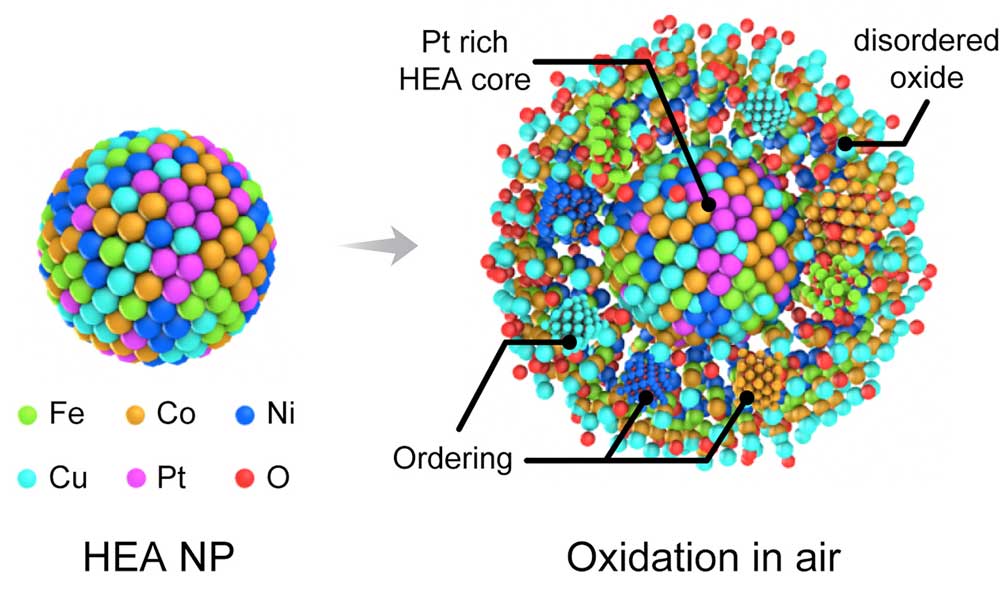
A new research report in ACS Nano (“In Situ Oxidation Studies of High-Entropy Alloy Nanoparticles”) offers key insights into how HEA nanoparticles behave under high-temperature oxidizing environment and sheds light on future design options of highly stable alloys under complex service conditions. This work has been a multidisciplinary and multi-institutional effort led by the University of Illinois at Chicago in collaboration with Argonne National Laboratory, University of Pittsburgh, University of California Riverside, and Northwestern University.”Traditional metals or alloys are believed to form crystalline oxides based on the composition and oxidation conditions, but when five or more principle elements are involved in a single phase, novel behavior during oxidation may exist due to the built-in high entropy,” Boao Song from the Department of Mechanical and Industrial Engineering at the University of Illinois at Chicago, and the paper’s first author, tells Nanowerk. “We were intrigued to find out that the transition metals in HEA nanoparticles can co-segregate into the oxide layer and form a disordered phase that can slow down the whole oxidation process.”
For the first time, this work directly captured the whole oxidation process of HEA nanoparticles in real-time and at the nanoscale. The advanced in situ transmission electron microscopy (TEM) technique used by the UIC team allows real-time imaging and analytical methods, and thus provides clear evidence of the co-segregation of transition metals during the oxidation process.
It also confirms the much slower oxidation kinetics of HEA nanoparticles. These findings suggest the possibility of utilizing these high-entropy alloys for applications where high thermal stability and oxidation resistance are required.The researchers managed to successfully capture the high-temperature oxidation of FeCoNiCuPt HEA nanoparticles in real-time by utilizing the advanced in situ gas-cell transmission electron microscopy (TEM) technique combined with density functional theory (DFT) calculations.
“We found that the oxidation kinetics of HEA nanoparticles are significantly slower than those of monometallic and bimetallic alloys,” says Song. “Combining in situ energy dispersive spectroscopy (EDS) and electron energy loss spectroscopy (EELS) analyses, we confirmed that the oxidation of HEA nanoparticles are governed by Kirkendall effects displaying outwards segregation of Fe, Co, Ni and Cu.”
“The present study is crucial for understanding HEA behavior in oxidizing environments and provides insights into designing high-temperature-resistant materials, durable catalysts, and corrosion-resistance alloys for various applications,”Song concludes. “Still, there are a lot of undiscovered phenomena with regards to HEAs and a lack of well-established theories to explain them. For example, the mechanism that controls the oxidation process and why the differences are so huge compared to monometallic nanoparticles is still unknown.”
So it appears that the team has their work cut out for them.
Researchers describe how quasiparticles called polarons behave in tellurene, a nanomaterial first synthesized in 2017 that is made up of tiny chains of tellurium atoms and has properties useful in sensing, electronic, optical and energy devices.
Read moreThe new method of making mixed halide-perovskites results in solar cells with improved stability and performance. The new method results in better control over perovskite crystallization rates. This means the crystal structure is more ordered, in part due to researchers understanding and taking advantage of the faster crystallization of bromide relative to iodide.
The result is a material with fewer defects and less halide migration and thus less segregation of the bromide and iodide. This in turn means uniform mixing of bromide and iodide across the material, which allows the material to absorb light evenly. The end result is that solar cells made using the new method will perform better under real-world conditions.
Typical halide perovskite solution deposition uses an anti-solvent drip procedure to initiate crystallization of the halide film. The standard anti-solvent method for producing bromide-iodide mixed halide perovskite films often leads to excessive defect formation (e.g., bromide vacancies) owing to the rapid crystallization of bromide vs. iodide-perovskite phases. Simulations show that halide migration is enhanced in the presence of a large population of halide vacancies. This limits the stability of bromide-iodide mixed halide perovskites under light and heat.
In comparison to the anti-solvent approach, the gentler gas-quench method better controls crystallization, first producing a bromide-rich surface layer that then induces top-down columnar growth to form a gradient structure with less bromide in the bulk than in the surface region. The anti-solvent method does not produce such a gradient structure.
In this study, researchers from the National Renewable Energy Laboratory, the University of Toledo, and the University of Colorado Boulder demonstrated that the gas-quench method also produces fewer bromide vacancies and results in materials with a higher quality opto-electronic performance. Solar cells made using the gas-quench method retain desirable light absorption properties and provide enhanced performance in the form of a higher charge carrier mobility, higher open circuit voltage, and enhanced stability.

